Our Story
Our Mission
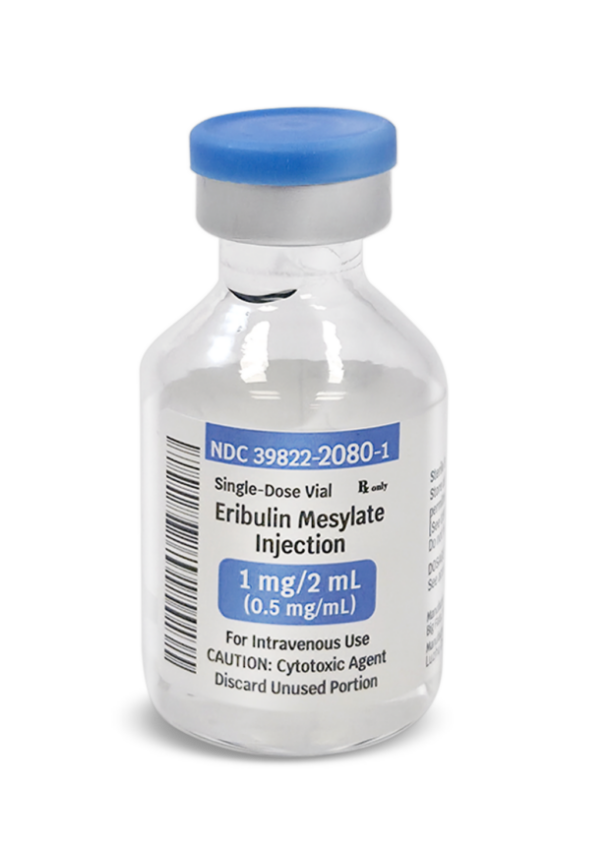
Eribulin Mesylate Injection
(10# NDC) 39822-2080-1
(11# NDC) 39822-2080-01
Disclaimer:
Product information on this website is intended for general information purposes only and is not a substitute for the approved product information. Buyers and consumers should not construe any information as medical advice or as a substitute for discussions with a qualified healthcare professional. XGen Pharmaceuticals DJB, Inc. and its affiliates accept no responsibility for the reliance on or use of any information contained herein.
1 mg / 2 mL (0.5 mg/mL) x 1 Vial
For Intravenous Use
Description:
Eribulin mesylate injection is a clear, colorless, sterile solution for intravenous administration. Each single-dose vial contains 1 mg of eribulin mesylate in 2 mL of solution. Each mL of solution contains 0.5 mg of eribulin mesylate (equivalent to 0.44 mg eribulin) in dehydrated alcohol (5% v/v) and water for injection (95% v/v). Sodium hydroxide or hydrochloric acid may be used for pH adjustment.
For more in-depth product and safety information, please review the full Prescribing Information or Packaging & Labeling links below.
Cencora (coming soon)
Cardinal (coming soon)
McKesson (coming soon)
Morris Dickson (coming soon)
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Do not freeze or refrigerate.
Store the vials in their original cartons.
For more in-depth product and safety information, please review the full Prescribing Information by following the link below.
Barcoded for Patient Safety.
Latex Free


XGEN PHARMACEUTICALS DJB, Inc. | 300 Daniel Zenker Drive | Horseheads NY, 14845
Our website uses session cookies.
View our Privacy Policy.